Biotherapies, or biomedicines, are defined by the way they are produced: a biotherapy is produced by a cell or a micro-organism, and is therefore bioproduced. As a result, the issues of production, safety and analysis of biomedicines are completely different from those of chemically synthesised small molecules. The emergence of different types of biotherapies has necessitated the introduction of new regulatory frameworks, as well as the creation of a fully-fledged biotherapies industry. In this highly innovative ecosystem, the classification of biomedicines is constantly evolving: at present, they are divided into four main categories, presented below.
The biologics biotherapies
Antibodies & Therapeutic proteins
They include all forms of protein: antibodies, recombinant proteins, peptidespeptides, etc.
Vaccines
They can be prophylactic or therapeutic. All types of vaccine (DNA, mRNA, inactivated, oncolytic, etc.) fall into this category.

Advanced therapy medicinal products (ATMPs)
They have emerged more recently than the therapeutic proteins. Cell & gene therapies (e.g. CAR-T cells) and tissue regeneration technology are all part of the ATMPs category.
The latest innovations
Some biotherapies don’t fit into the above three categories, but the therapeutic hope they represent is very strong: for example, microbial therapy, such as antibacterial phage therapy.
A new market, new challenges
Compared to small chemical molecules, biopharmaceuticals are a relatively recent class of molecules: in 2022, they accounted for just 4% of marketed drugs.
However, the market for biologics is growing rapidly, and is expected to reach $600 billion by 2026. This trend is illustrated by a 64% increase in the number of biopharmaceuticals under development worldwide between 2019 and 2022. In 2025, biologics account for 61% of products under development
Biologics have opened up new prospects, representing a market of the future and a hope for the treatment of many diseases.
of molecules under development worldwide are biopharmaceuticals

23 297 products

6 496 companies

Despite the small number of molecules on the market, biopharmaceuticals account for 32% of drug sales: this high proportion in terms of sales is explained by higher sales prices than for small molecules.
With the coming boom in biopharmaceuticals to treat an ever-increasing number of pathologies, these costs will be unsustainable for healthcare systems. The need to reduce development and production costs for biologics makes biomanufacturing a major priority for the sector.
The French industry
As an association of the French biopharmaceutical industry, MabDesign aims to structure the biotherapies and biomanufacturing sector, from R&D through biomanufacturing to market launch. We therefore list the companies and individuals involved in the sector as exhaustively and accurately as possible
The French biopharmaceutical ecosystem is a rich and innovative one, with more than a thousand structures, ranging from research institutions to biotechnology companies. The players in the biotherapies sector are spread across the whole of France, with two key regions for historical reasons: Île-de-France and Auvergne-Rhônes-Alpes.
The main challenge facing the biotherapies sector is to meet future needs in an innovative and fast-growing sector that is constantly evolving.
1285 structures in France
Scroll over the regions to see the distribution of stakeholders (%)
The French industry includes The French industry includes 271 biotechs/pharma, developing biologics. These companies are supported by 823 companies offering a full range of services : suppliers, bioanalysis, CRO, CDMO, etc.
The french biologics pipeline is rich, with 904 projects in development, 48% of which are currently in clinical phase. In terms of product types, French biotechs are developing products across the entire spectrum of biotherapeutics, with antibodies and MTIs leading the way. In terms of product types, French biotechs are developing products across the entire spectrum of biotherapeutics, with antibodies and MTIs leading the way.
Mabdesign, LEEM and FranceBioLead have joined forces to set up a tool to identify biotherapy industry players based in France, which can be consulted online by anyone: MyBiologicsPartner. It facilitates collaboration between players in the industry, with directories dedicated to prophylactic vaccines, recombinant and other protein antibodies, cell therapies, gene therapies and CAR-T cells. It facilitates collaboration between players in the industry, with directories dedicated to prophylactic vaccines, recombinant and other protein antibodies, cell therapies, gene therapies and CAR-T cells.
Biotherapies pipeline in France : 904 projects and 542 unique products in development
The European biologics sector
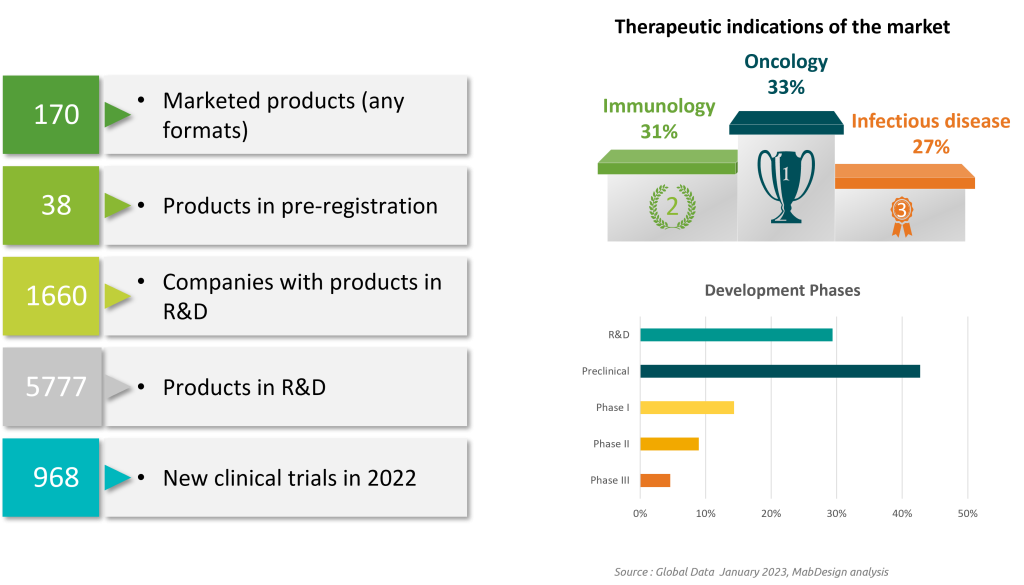
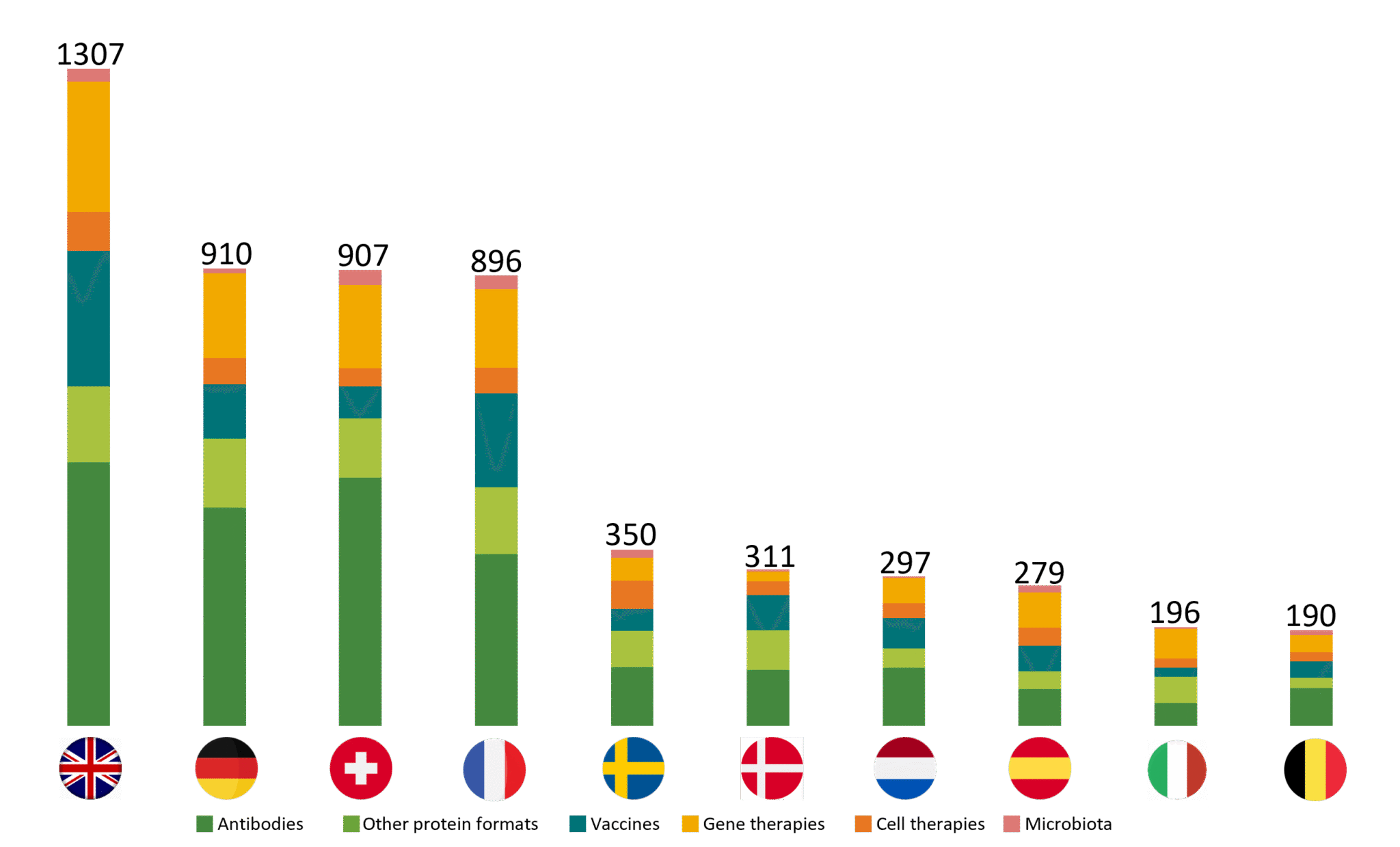
European biotechs develop 16 % of worldwide biotherapies, compared to 43 % for North America. The European ecosystem is driven by 4 leading countries: the UK, Germany, Switzerland and France.
Therapeutic antibody and protein formats account for the majority of products under development. As with vaccines and cell therapies, the majority of products are in clinical phases. Gene therapies, a less mature technology accounting for 18% of products in development, are three-quarters in pre-clinical or discovery development.
Biotherapies pipeline in Europe : 6022 projects, and 3729 unique products in development.
Biopharmaceuticals in development (top 10 countries, projects)
Last data update : October 2025