MabDesign’s market analyses give you a monthly overview of market dynamics and projects in development for a given indication or technology, to help you better understand the constantly evolving market of biologics and biomanufacturing. This month, an overview of the market and pipeline of cell therapies.
Market & pipeline analysis
Cell therapies continue to expand as a major pillar of advanced therapeutic innovation. Initially driven by autologous approaches, the field now encompasses a broad spectrum of cell sources, therapeutic strategies and technological platforms. The market is progressively maturing, while the development pipeline reflects strong diversification and sustained R&D activity.
Key figures
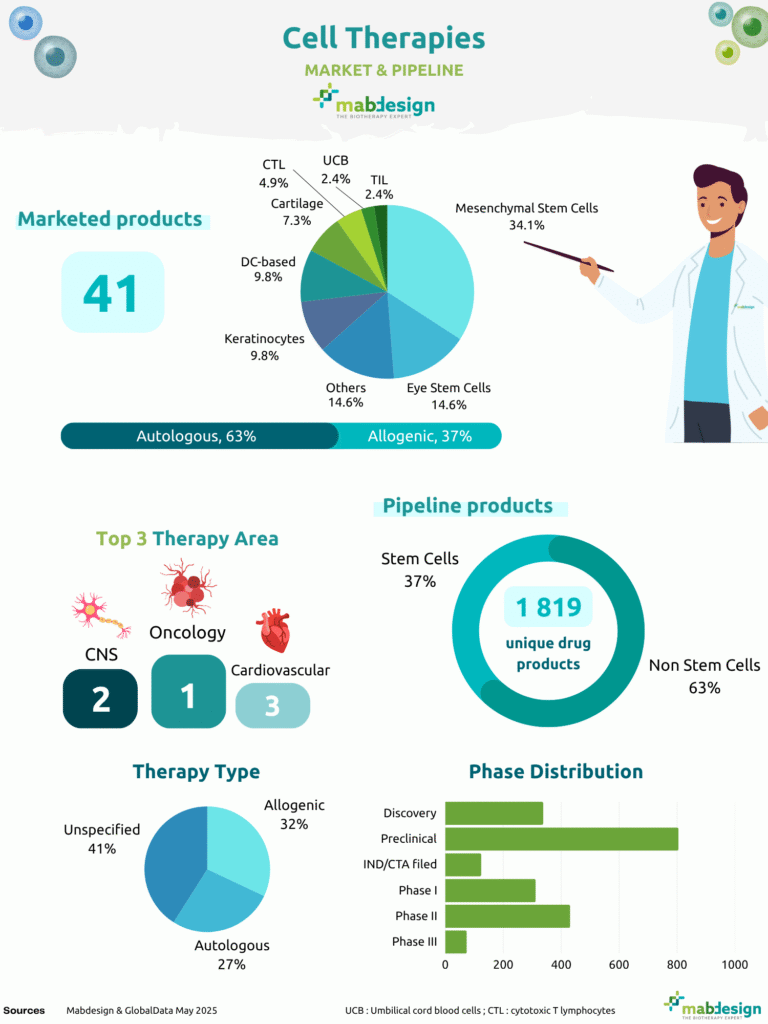
41
Marketed cell therapy products
63%
Autologous products
37%
Allogeneic products
To date, 41 cell therapy products are marketed worldwide. Autologous approaches remain predominant, accounting for 63% of approved products, reflecting historical development pathways and established regulatory precedents. Allogeneic products represent 37% of marketed therapies, illustrating the gradual shift toward scalable and off-the-shelf strategies.
Current trends
Marketed product landscape by cell type
Among approved cell therapies, mesenchymal stem cells account for the largest segment (34.1%), confirming their central role in early clinical translation and commercialisation. Eye stem cells and other cell types each represent 14.6% of marketed products, followed by keratinocytes and DC-based therapies (9.8% respectively), while cartilage-derived therapies account for 7.3%.
More specialised immune cell approaches remain marginal in the marketed landscape, with CTL representing 4.9% and both TIL and umbilical cord blood (UCB)–based therapies accounting for 2.4% each. Overall, this distribution reflects the early clinical success of stem cell–based strategies, particularly mesenchymal stem cells, across regenerative and inflammatory indications.
Active pipeline and phase distribution
The global cell therapy pipeline currently comprises 1,819 unique drug products, reflecting sustained R&D activity and continued diversification of therapeutic strategies. While stem cell–based approaches remain significant, the development landscape is increasingly driven by alternative cell platforms and engineered strategies.
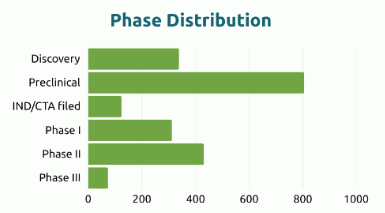
63%
Non-stem cell approaches
37%
Stem cell–based approaches
Beyond format diversification, the phase distribution highlights the developmental maturity of the field. The majority of candidates remain concentrated in preclinical stages, followed by assets in Phase I and Phase II, while only a limited number have progressed to Phase III. This structure underscores both the intensity of early-stage innovation and the scientific, manufacturing and regulatory challenges that continue to shape the progression of cell therapy programs toward late-stage development and commercialisation.
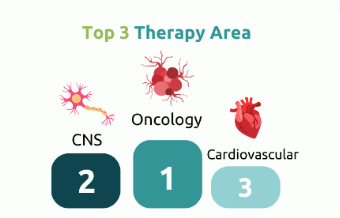
Therapy areas and therapy type
The cell therapy pipeline is primarily driven by oncology, which remains the leading therapeutic area in terms of development activity. This predominance reflects the continued expansion of immune cell–based approaches and cellular strategies in cancer treatment. Beyond oncology, the central nervous system (CNS) and cardiovascular diseases represent the second and third most targeted areas, highlighting growing interest in regenerative medicine and cell-based tissue repair applications.
In parallel, the distribution by therapy type illustrates the strategic orientation of development programs:
- Unspecified: 41%
- Allogeneic: 32%
- Autologous: 27%
Compared with the marketed landscape, the pipeline shows a more balanced distribution between autologous and allogeneic approaches considering that 40% of the drug candidates are not yet specified. The increasing share of allogeneic programs reflects industrial efforts to enhance scalability, manufacturing standardisation and broader patient accessibility, which remain key determinants for the long-term sustainability of cell therapy platforms.
Conclusion
Cell therapies represent a rapidly evolving segment of the advanced therapeutics landscape. While marketed products remain limited in number and largely autologous, the pipeline reflects strong diversification, with growing emphasis on allogeneic and non-stem cell approaches. As manufacturing capabilities and regulatory frameworks mature, the coming years will be critical in determining the scalability and commercial sustainability of next-generation cell therapies.
Download our infographic
projets de thérapies basées sur l’édition du génome (gene-editing therapies) actuellement en développement. (2025)
Want to know more?
MabDesign has acquired and cultivated expertise in all market segments associated with biopharmaceuticals and biomanufacturing. Our team of consultants is available to assist you with all your biotherapy-related projects. Feel free to explore our range of services, including our marketing and strategic analyses, if you would like to learn more!
Sources : MabDesign & GlobalData
Date – Août 2025